Research Summary
Our lab is interested in understanding how collective cell migration and tissue mechanics contributes to intestinal function and disease. The mammalian intestine is well-organized and highly dynamic tissue, with the vast majority of cells exhibiting rapid collective migration and turnover. We are particularly interested in understanding how cell mechanics and physical interactions between neighboring cells and their substrate affect collective migration dynamics and how migration patterns and cell turnover in turn affect tissue function. To address these questions, our lab employs a multidisciplinary approach, combining cell biology and live imaging with quantitative analytical methods, biophysics and theoretical modeling.
Background
The small intestine is segregated into stereotyped patterns of crypts and villi. Crypts contain stem and progenitor cells that, upon differentiation, migrate toward the villus tip and are extruded, turning over every 3-5 days, all while maintaining a tight epithelial barrier. Defects in barrier function is associated with inflammatory bowel disease (IBD), a major risk factor for intestinal cancer. IBD is characterized by poor epithelial barrier function and chronic inflammation and is influenced by the gut microbiota. We are interested in better understanding how cell patterning and collective migration contribute to maintaining this tissue barrier, especially in inflammatory conditions.
Ongoing Research
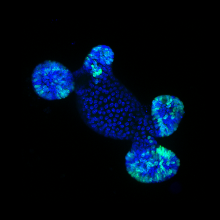
To study intestinal cell and tissue function, we primarily use intestinal organoids grown in 3D or 2D environments. Organoids recapitulate many aspects of the in vivo gut, including cell type compartmentalization, tissue folding and geometry and collective migration. Organoids are a model uniquely suited to study many aspects of epithelial and intestinal homeostasis such as stem cell renewal, differentiation and wound healing as well as interactions with the immune system and microbiome. In the lab, we employ state-of-the-art culturing and microfabrication techniques to better understand these processes and how individual cell function contributes to overall tissue health and disease.
Additional Lab Info
The Clark research group is funded as part of the Excellence Strategy of the German Federal and State Governments at the Univeristy of Tübingen. Our lab is also affiliated with the Stuttgart Research Center Systems Biology (SRCSB) and the Center for Personalized Medicine at the University of Tübingen (ZPM). For more details on past and present research from the lab as well as additional resources, feel free to check out Andrew’s website.
Lab Members
Andrew Clark
Hoang Trinh Thao Nguyen (Postdoc)
Sarbari Saha (PhD Student)
Fabian Gärtner (Bachelor Student)
Open positions
We are happy to support highly qualified and competitive candidates that wish to apply for fellowships or external funding to join our research team. Open calls for funded positions in the lab will be advertised on this page and stellenwerk-stuttgart.de. We are also happy to host Bachelor and Master students in the lab. Please contact Andrew with your CV and research interests if you are interested.
Selected publications
Pérez-González C, Ceada G, Greco F, Matejčić M, Gómez-González M, Castro N, Menendez A, Kale S, Krndija D, Clark AG, Gannavarapu VR, Álvarez-Varela A, Roca-Cusachs P, Batlle E, Vignjevic DM, Arroyo M and Trepat X (2021) Mechanical compartmentalization of the intestinal organoid enables crypt folding and collective cell migration. Nature Cell Biology 23, 745–757. https://www.nature.com/articles/s41556-021-00699-6
Clark AG, Maitra A, Jacques C, Simon A, Pérez-González C, Trepat X, Voituriez R and Vignjevic DM (2020) Viscoelastic relaxation of collagen networks provides a self-generated directional cue during collective migration. biorXiv. https://doi.org/10.1101/2020.07.11.198739
Clark, A.G. (2021) Biophysical origins of viscoelasticity during collective cell migration. In: Pajic-Lijakovic, I. and Barriga, E.H. Viscoelasticity and Collective Cell Migration: An interdisciplinary perspective across levels of organization. Elsevier/Academic Press, Cambridge, MA, USA.https://doi.org/10.1016/B978-0-12-820310-1.00007-0
Staneva, R., El Marjou, F., Barbazan, J., Krndija, D., Richon, S., Clark, A.G.*, and Vignjevic, D.M.* (2019) Cancer Cells in the Tumor Core Exhibit Spatially Coordinated Migration Patterns. J Cell Sci, 132(6):jcs220277. https://doi.org/10.1242/jcs.220277
Chugh, P.*, Clark, A.G.*, Smith, M.B.*, Cassani, D.A.D., Ragab, A., Roux, P.P., Charras, G., Salbreux, G. and Paluch, E.K. (2017) Actin Cortex Architecture Regulates Cell Surface Tension. Nat Cell Biol, 19(6):689-697. https://dx.doi.org/10.1038/ncb3525
Aizel, K.*, Clark, A.G.*, Simon, A., Geraldo, S., Funfak, A., Vargas, P., Bibette, J., Vignjevic, D.M. and Bremond, N. (2017) A Tuneable Microfluidic System for Long Duration Chemotaxis Experiments in a 3D Collagen Matrix. Lab Chip, 17(22):3851-3861. https://dx.doi.org/10.1039/C7LC00649G
Clark, A.G., and D.M. Vignjevic (2015) Modes of Cancer Cell Invasion and the Role of the Microenvironment. Current Opinion in Cell Biology. 36:13-22. https://doi.org/10.1016/j.ceb.2015.06.004