Research overview
Cancer development is accompanied by the generation of tumour evasion mechanisms from the immune system. The challenge of cancer immunotherapy consists in enabling the immune system again to recognize and eliminate the tumour cells. A key feature is to overcome the immunosuppressive conditions in the tumour microenvironment. This can be achieved by interfering with the complex regulatory network of the immune system, shifting the balance towards supporting and driving an anti-tumour response. Here, in particular members of the B7/CD28-superfamily, TNF/TNFR-superfamily and cytokines/common γ-chain receptor family have shown great potential. We are interested in exploring how tumour-directed delivery and defined combinations of such immunomodulatory molecules can enhance the immune response in the tumour microenvironment leading to novel therapeutic strategies.
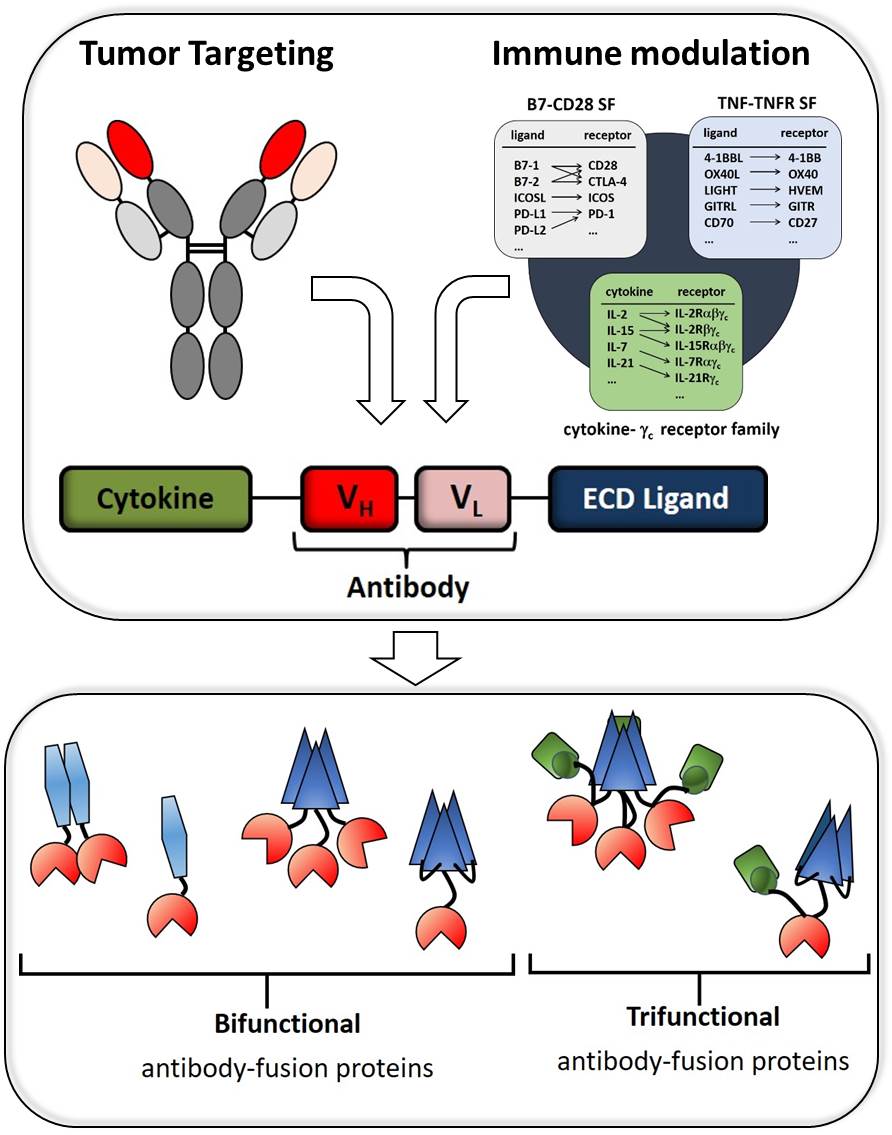
Targeted strategies with bi- and trifunctional antibody-fusion proteins
Tumour targeting can be achieved by antibodies binding specifically to tumour associated antigens (TAA). This makes recombinant antibodies a valuable tool for diverse targeted approaches. For example, bispecific antibodies (TAAxCD3) can retarget effector cells to tumour cells, inducing artificially an antitumor immune response. Also, ligands or cytokines fused to recombinant antibodies can be directed to and presented at the tumour cell surface to immune cells, modulating their response. Certainly, both approaches can also be combined. In order to explore the potential and improve the efficacy of such molecules we design different formats of bi- and trifunctional antibody-fusion proteins, focusing on optimized presentation and a combined mode of action. Analysis are carried out in 2D- and 3D-coculture settings with tumour cells/immune cells in vitro and in tumour mouse models in vivo.
Publications
Selected research articles
- Möller AM, Vettermann S, Baumann F, Pütter M, Müller D. (2025) Trifunctional antibody-cytokine fusion protein formats for tumor-targeted combination of IL-15 with IL-7 or IL-21. Front Immunol. 16:1498697. doi: 10.3389/fimmu.2025.1498697.
- Sapski S, Beha N, Kontermann R, Müller D. (2020) Influence of antigen density and immunosuppressive factors on tumor-targeted costimulation with antibody-fusion proteins and bispecific antibody-mediated T cell response. Cancer Immunol Immunother. 69(11):2291-2303. doi: 10.1007/s00262-020-02624-6.
- Beha N, Harder M, Ring S, Kontermann RE, Müller D. (2019) IL15-Based Trifunctional Antibody-Fusion Proteins with Costimulatory TNF-Superfamily Ligands in the Single-Chain Format for Cancer Immunotherapy. Mol Cancer Ther. 18(7):1278-1288. doi: 10.1158/1535-7163.MCT-18-1204.
- Fellermeier-Kopf S, Gieseke F, Sahin U, Müller D, Pfizenmaier K, Kontermann RE. (2018) Duokines: a novel class of dual-acting co-stimulatory molecules acting in cis or trans. Oncoimmunology. 7(9):e1471442. doi: 10.1080/2162402X.2018.1471442.
- Sapski S, Beha N, Kontermann R, Müller D. (2017) Tumor-targeted costimulation with antibody-fusion protein improves bispecific antibody-mediated immune response in presence of immunosuppressive factors. OncoImmunology 6(12):e1361594. doi: 10.1080/2162402X.2017.1361594.
- Fellermeier S, Beha N, Meyer JE, Ring S, Bader D, Kontermann RE, Müller D (2016) Advancing targeted costimulation with antibody-fusion proteins by introducing TNF superfamily members in a single-chain format. OncoImmunology 5(11):e1238540. doi: 10.1080/2162402X.2016.1238540.
- Kermer V, Hornig N, Harder M, Bondarieva A, Kontermann RE, Muller D. (2014) Combining antibody-directed presentation of IL-15 and 4-1BBL in a trifunctional fusion protein for cancer immunotherapy.Mol Cancer Ther. 13(1):112-21. doi: 10.1158/1535-7163.MCT-13-0282.
- Hornig N, Reinhardt K, Kermer V, Kontermann RE, Müller D. (2013) Evaluating combinations of costimulatory antibody-ligand fusion proteins for targeted cancer immunotherapy.Cancer Immunol Immunother. 62(8):1369-1380. doi: 10.1007/s00262-013-1441-7.
- Hornig N, Kermer V, Frey K, Diebolder P, Kontermann RE, Müller D. (2012) Combination of a bispecific antibody and costimulatory antibody-ligand fusion proteins for targeted cancer immunotherapy.J Immunother. 35(5):418-29. doi: 10.1097/CJI.0b013e3182594387.
- Kermer V, Baum V, Hornig N, Kontermann RE, Müller D. (2012) An Antibody Fusion Protein for Cancer Immunotherapy Mimicking IL-15 trans-Presentation at the Tumor Site. Mol Cancer Ther 11: 1279-1288. doi: 10.1158/1535-7163.MCT-12-0019.
- Müller D, Frey K, Kontermann RE (2008) A novel antibody-4-1BBL fusion protein for targeted costimulation in cancer immunotherapy. J Immunother. 31(8):714-22. doi: 10.1097/CJI.0b013e31818353e9.
Review articles
- Müller D (2023) Targeting Co-Stimulatory Receptors of the TNF Superfamily for Cancer Immunotherapy. BioDrugs. 37(1):21-33. doi: 10.1007/s40259-022-00573-3.
- Müller D (2015) Antibody fusions with immunomodulatory proteins for cancer therapy.Pharmacol Ther. 154:57-66. doi: 10.1016/j.pharmthera.2015.07.001.
- Müller D (2014) Antibody fusion proteins for cancer immunotherapy: an update on recent developments. BioDrugs. 28(2):123-31. doi: 10.1007/s40259-013-0069-7.
- Müller D (2012) Targeted cancer immunotherapy: mimicking physiological trans-presentation of IL-15. OncoImmunology 1(7):1213-1214. doi: 10.4161/onci.20824.
- Müller D, Kontermann RE (2010) Bispecific antibodies for cancer immunotherapy: Current perspectives. 24(2):89-98. doi: 10.2165/11530960-000000000-00000.
- Muller D, Kontermann RE (2007) Recombinant bispecific antibodies for cellular cancer immunotherapy. Curr Opin Mol Ther. 9(4):319-26.
Book chapters
- Siegemund M, Beha N, Müller D. (2018) Production, Purification, and Characterization of Antibody-TNF Superfamily Ligand Fusion Proteins.In: Antibody Engineering, Methods and Protocols. 3th Nevoltris and P. Chames (Eds.), Springer Nature, New York, pp.351-364.
- Müller, D. and Kontermann, R.E. (2014) Bispecific Antibodies In: Handbook of Therapeutic Antibodies. 2thDübel S. (Hrsg.), WILEY-VCH Verlag, Weinheim. Vol.I pp.267-293
- Müller, D. & Kontermann, R.E. (2011) Recombinant bispecific antibodies for cancer therapy. In: Antibody Expression and Production (Cell Engineering 7), Al-Rubeai M. (Ed.), Springer-Verlag, Heidelberg, pp.235-250.
- Müller, D. & Kontermann, R.E. (2011) Diabodies, single-chain diabodies and their derivatives. In: „Bispecific Antibodies“, Kontermann R.E. (Ed.), Springer-Verlag, Heidelberg, pp. 83-100.
- Müller D & Gerspach J (2010) Antibody-Cytokine Fusion Proteins with Members of the TNF-Family. In: Antibody Engineering R Kontermann and S. Dübel (eds.) Springer-Verlag Berlin Heidelberg. 2 pp. 113-126.
Lab members
PD Dr. Dafne Müller (PI)
Annika Möller (PhD student)
Open positions
Applications of students and scientists interested in biomedical engineering and cancer immunotherapy are always welcome! Please send your CV and a short summary of your research interests to ![]() Dr. Dafne Müller
Dr. Dafne Müller
Funding
Our group has received funding from: DFG, German Cancer Aid and industry collaboration.